What is circadian medicine?
Why when you treat matters as much as what you treat
Modern medicine is precise about dosage. It is precise about diagnosis. It is increasingly precise about genetics. But it remains remarkably imprecise about timing — and that oversight may be costing patients outcomes that are already within reach.
Your body doesn't run on clock time
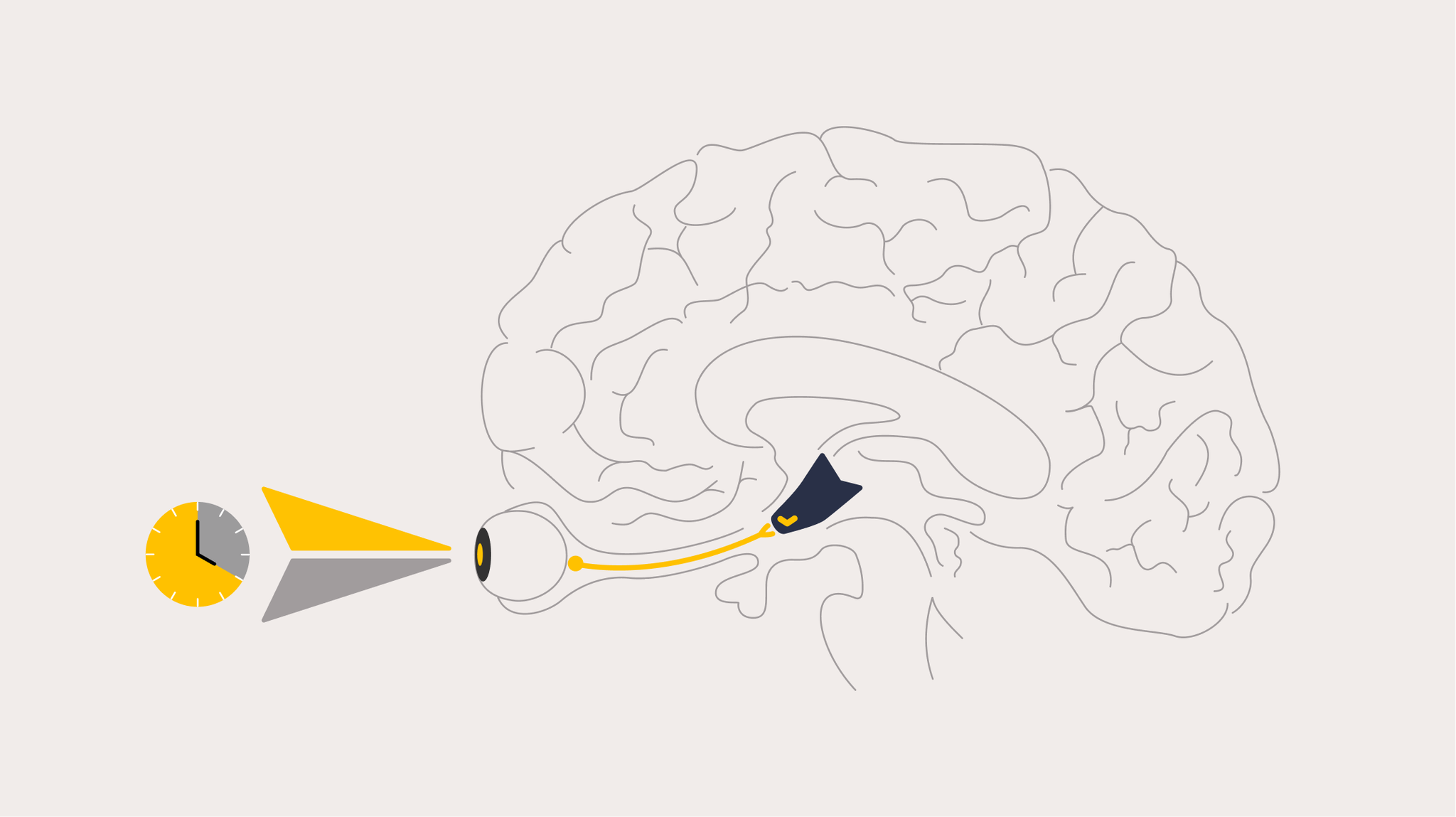
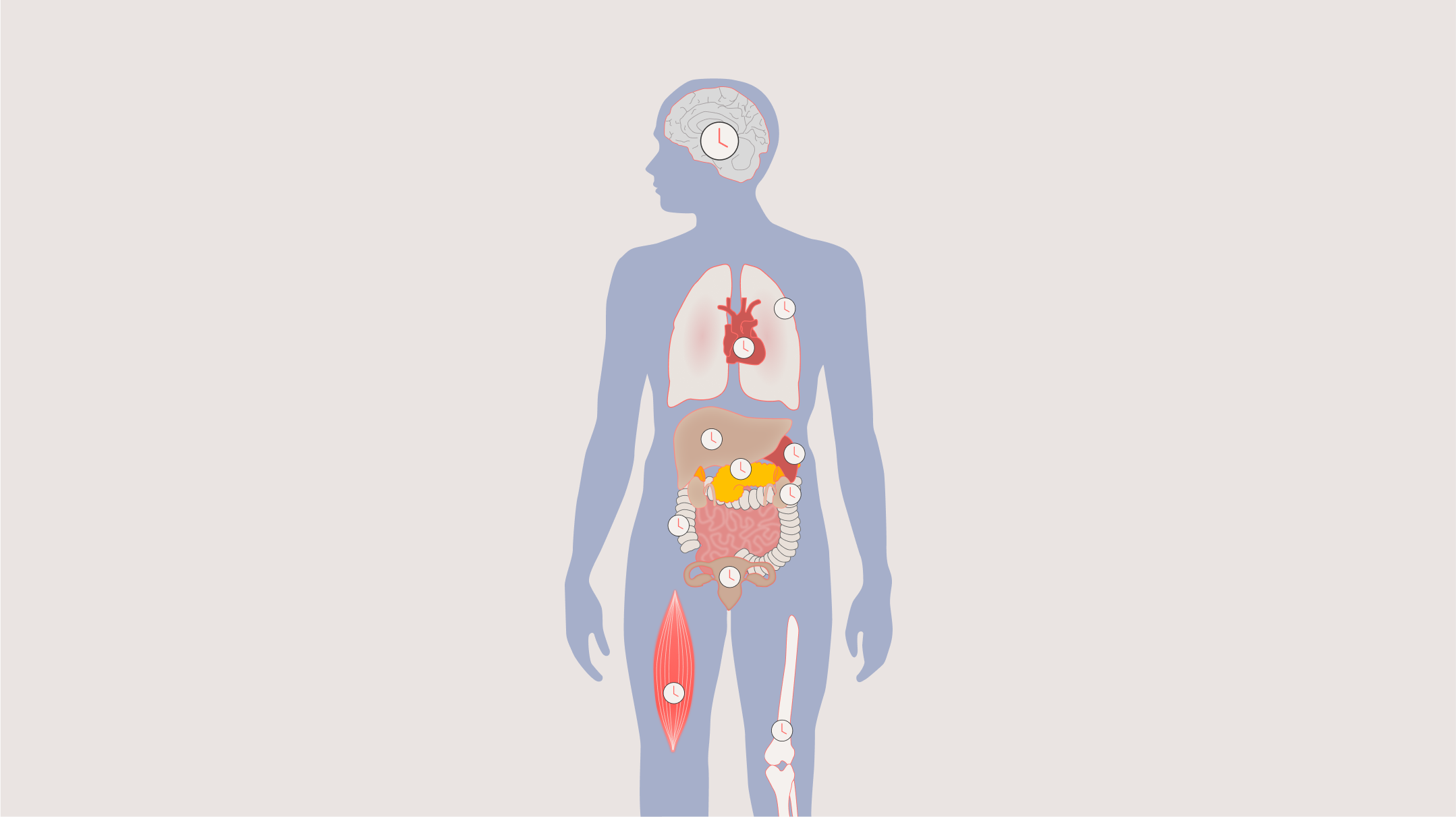
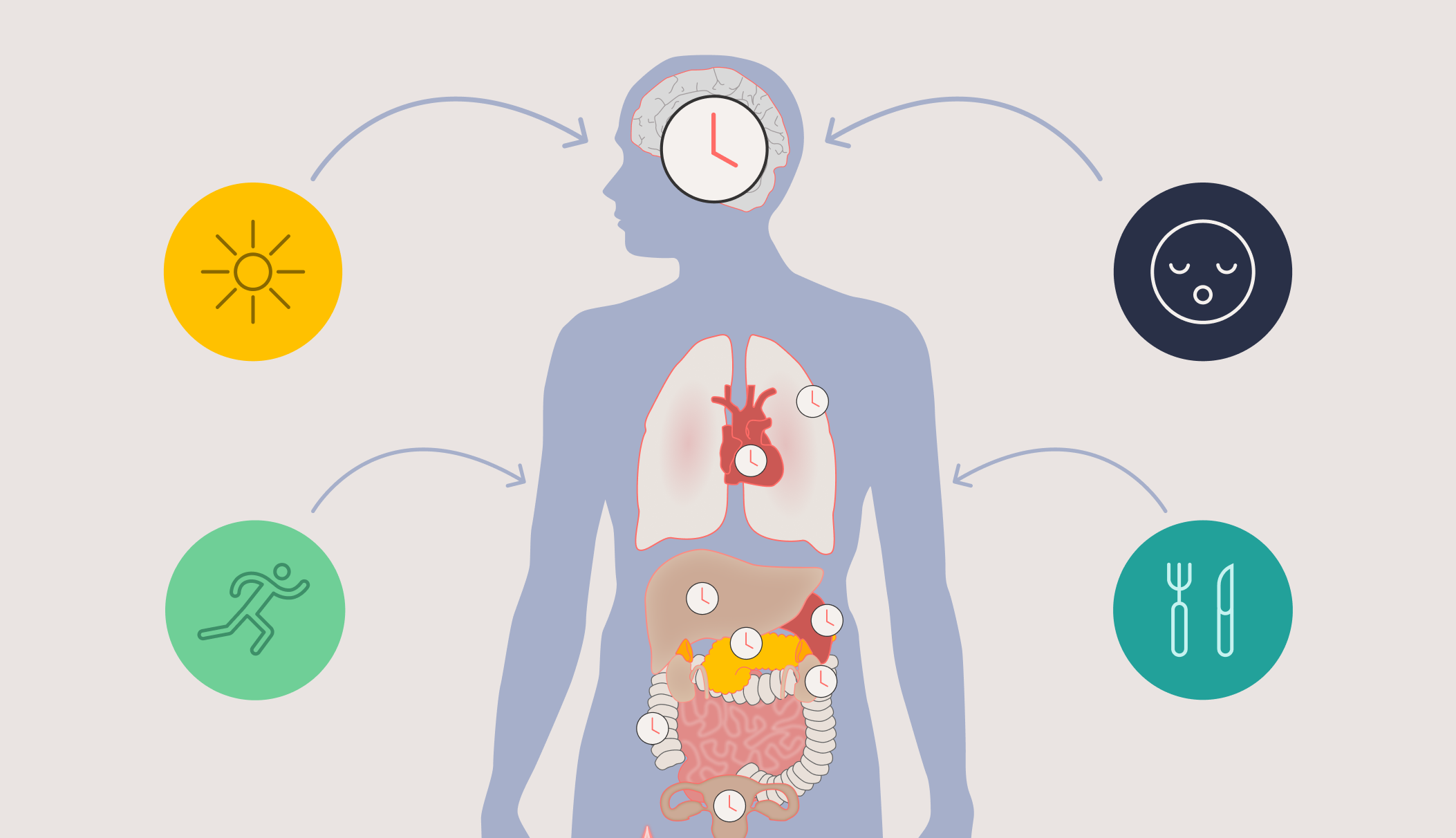
The circadian system — driven by the suprachiasmatic nucleus (SCN), a master clock in the brain — orchestrates near-24-hour rhythms across virtually every organ and biological process in the body. Heart function, liver metabolism, immune response, lung capacity, glucose regulation, drug metabolism, mood, and performance all rise and fall in predictable daily cycles.
But here's the critical insight: these rhythms don't follow the clock on the wall. They follow each individual's internal circadian time — and that varies enormously from person to person. Research has shown that even under highly controlled laboratory conditions, the timing of melatonin onset (a reliable marker of circadian phase) varies by five to six hours between individuals (Sletten et al., *PLoS One*, 2015). In real-world conditions, that range widens dramatically — up to nine hours in university students (Phillips et al., *Scientific Reports*, 2017) and twelve hours in shift workers (Dumont et al., *Journal of Biological Rhythms*, 2001).
This means that two patients receiving the same medication at the same clock time — say, 8 a.m. — may be at completely different points in their circadian cycle. One may be receiving the drug near peak efficacy. The other may be missing that window entirely.
Dosing time changes outcomes
The idea that timing matters in medicine isn't speculative. A landmark review by Ruben et al. published in *Science* (2019) analyzed 106 published drug studies across 15 therapeutic areas and found that in 75% of cases, treatment efficacy or toxicity depended on dosing time. For drugs with half-lives under 15 hours, that figure rose to 85%.
The conditions affected span some of medicine's most prevalent and costly challenges: hypertension, cancer, asthma, arthritis. Multiple clinical trials have demonstrated that radiation therapy produces fewer side effects when administered at specific times of day — likely because healthy cells are more resistant to damage at certain circadian phases. And research on vaccine timing has shown that the time of day a vaccine is administered can significantly influence the strength of the immune response (De Bree et al., *Journal of Clinical Investigation*, 2020).
Yet for the vast majority of approved drugs, the impact of dosing time has never been explored.
From clock time to circadian time
Most existing research on treatment timing has used clock time — giving patients a drug at, say, 8 a.m. versus 8 p.m. This approach shows promise, but it has a fundamental limitation: it treats all patients as if their internal clocks are synchronized. They are not.
Circadian medicine takes this a step further. Rather than prescribing based on the time on the wall, it aims to time treatments to each patient's individual circadian phase — the point in their personal biological cycle when a drug, vaccine, or therapy would be most effective or least toxic.
This requires two capabilities: accurately determining an individual's circadian phase (using biomarkers such as dim light melatonin onset, or DLMO), and then either timing the treatment to match that phase — or, when a treatment time is fixed, shifting the patient's circadian clock so that their biology aligns with the schedule.
Why this matters now
The implications for both approved drugs and drugs in development are substantial. For existing medications, circadian-informed dosing could improve efficacy and reduce side effects without changing the drug itself — offering competitive differentiation, extended patent life, and better patient outcomes. For drugs in clinical trials, incorporating circadian timing could reduce toxicity-related dropouts, sharpen results, and increase the likelihood of regulatory approval.
As Ruben et al. noted in their *Science* review, this knowledge alone could be the determining factor in whether a drug receives FDA approval.
The next frontier in personalized medicine
Precision medicine has focused largely on *what* — the right drug for the right patient based on their genetics, biomarkers, and disease profile. Circadian medicine adds a new dimension: *when*. It recognizes that the human body is not a static system but a dynamic one, with biological processes that peak and trough in rhythms that vary from person to person.
Timeshifter is pioneering this frontier — translating decades of circadian neuroscience into tools that make personalized circadian timing actionable at scale. The same science that helps astronauts, elite athletes, and shift workers perform at their best is now being applied to optimize clinical outcomes.
The clock is already ticking. The question is whether medicine will start listening to it.
References
- De Bree et al. (2020) — Non-specific effects of vaccines: current evidence and potential implications. J Clin Invest.
- Dumont et al. (2001) — Profile of 24-h light exposure and circadian phase of melatonin secretion in night workers. J Biol Rhythms.
- Flynn-Evans et al. (2017) — Circadian phase and phase angle disorders in primary insomnia. Sleep.
- Phillips et al. (2017) — Irregular sleep/wake patterns are associated with poorer academic performance and delayed circadian and sleep/wake timing. Sci Rep.
- Ruben et al. (2019) — Dosing time matters. Science.
- Ruben et al. (2019) — A large-scale study reveals 24-hr operational rhythms in hospital treatment. PNAS.
- Sletten et al. (2015) — Inter-individual differences in neurobehavioural impairment following sleep restriction are associated with circadian rhythm phase. PLoS One.
- Van Diemen et al. (2020) — Chronotherapy of conventional low-dose aspirin in healthy volunteers. Platelets.